AI-Powered Drug Discovery: Accelerating Solutions to Complex Biomedical Challenges
Breaking the limits of traditional drug discovery with AI-driven speed, precision, and scalability.
Introduction
In the realm of modern biotechnology, the pace of scientific discovery has traditionally been constrained by the labor-intensive processes of hypothesis generation, experimental design, and data analysis. However, the advent of artificial intelligence (AI) has reshaped these paradigms, enabling researchers to tackle intricate molecular challenges with unprecedented speed and precision. One pivotal application of AI in biotechnology is drug discovery—a field known for its complexity, cost-intensive workflows, and long timelines. In this article, we delve into how AI, computational chemistry, and enzyme engineering are spearheading innovation in the biopharmaceutical sector, with a focus on how platforms like Medvolt facilitate breakthroughs.
Understanding AI-Driven Drug Discovery
Conventional drug discovery relies on iterative trial-and-error processes to identify therapeutic candidates, optimize molecular profiles, and ensure efficacy and safety. While necessary, this approach has significant downsides: a high attrition rate, lengthy timelines (often 10–15 years), and substantial costs, averaging over $2.5 billion per drug. AI-driven methodologies are transforming these constraints by enabling researchers to:
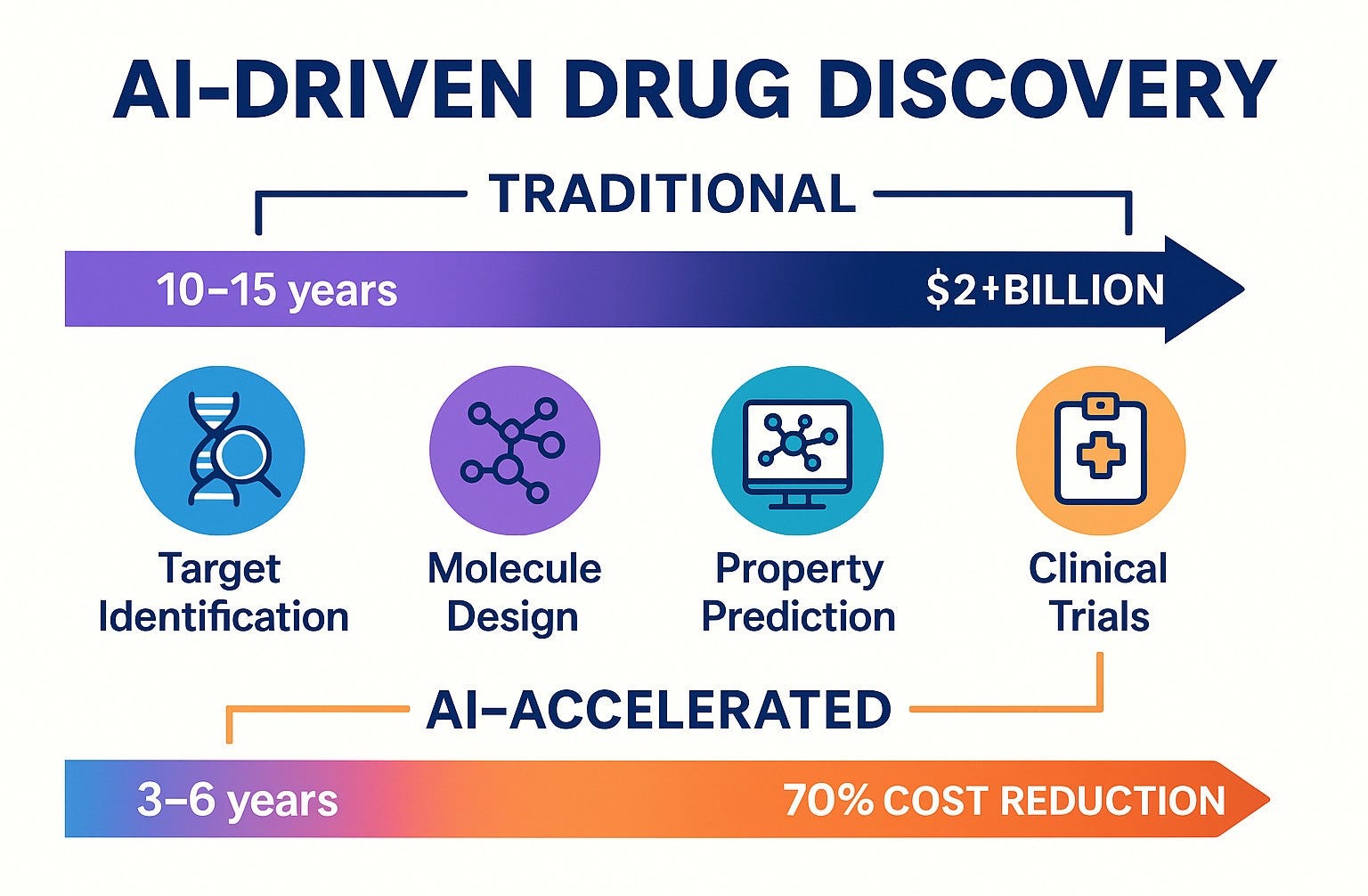
- Predict molecular efficacy and safety using machine learning models trained on multi-omics datasets.
- Perform high-throughput screening with computational tools such as fragment-based molecular design.
- Simulate molecular dynamics via tools like free energy perturbation (FEP) calculations.
- Optimize lead compounds by integrating data from virtual libraries, cheminformatics, and biological assays.
The result? A paradigm shift where drug discovery timelines are compressed, screening is more targeted, and candidate therapies are prioritized for clinical success.
Computational Chemistry: Crafting Precision Therapies
Computational chemistry lies at the heart of AI-assisted drug discovery, offering atomistic insights into molecular interactions. Techniques such as molecular modeling, docking simulations, and quantum mechanics/molecular mechanics (QM/MM) hybrid methods are enabling precision in identifying drug candidates with optimal binding affinities and specificity.
Molecular Docking and Virtual Screening
Virtual screening leverages AI models to analyze millions of molecular fragments against specific biological targets, effectively narrowing down the pool of candidates in silico before experimental validation. Molecular docking algorithms, for instance, assess binding poses of compounds within active sites, optimizing parameters that determine efficacy.
Free Energy Perturbation (FEP) Simulations
FEP simulations offer unparalleled accuracy in quantifying the binding free energy between drug molecules and protein targets—a critical metric for predicting therapeutic potential. By incorporating AI into these simulations, researchers accelerate high-fidelity insights, bridging the gap between theoretical chemistry and experimental outcomes.
Multi-Omics Integration for Biomarker Discovery
A major bottleneck in drug research is understanding disease mechanisms across complex biological systems. Multi-omics provides a solution, integrating genomics (DNA), transcriptomics (RNA), proteomics (proteins), and metabolomics (small molecules) data to uncover disease biomarkers and therapeutic pathways. AI platforms streamline multi-omics analysis by identifying patterns and correlations buried in massive, heterogeneous datasets.
Enzyme Engineering Applications
In addition to drug discovery, advances in AI-driven enzyme engineering are proving transformative for the biomanufacturing sector. Rational enzyme design, powered by AI and structural biology tools, optimizes catalytic activity, stability, and substrate specificity, enhancing industrial workflows from green chemistry applications to synthetic biology products. By combining sequence-function mapping with computational libraries, researchers create tailored biocatalysts for drug synthesis, minimizing waste and reaction times.
Medvolt’s Role in Innovation
Medvolt exemplifies how AI and computational platforms are driving drug discovery forward. Through the integration of molecular modeling, FEP simulations, and knowledge curation systems, Medvolt’s technologies enable life sciences teams to reduce drug development timelines while navigating cost-efficient strategies for precision medicine.
Use Case: Accelerating Oncology Drug Therapies
One of Medvolt’s notable contributions is in oncology drug research, where traditional methods grapple with dynamic resistance mechanisms in cancer cells. By employing AI-driven molecular dynamic simulations, Medvolt platforms have helped identify compounds targeting driver mutations in cancer pathways. These insights feed into rational design workflows, helping develop therapies with high specificity and reduced off-target effects.
Predictive Algorithms for Toxicity and ADMET Profiling
Medvolt’s algorithms also offer predictive frameworks for absorption, distribution, metabolism, excretion, and toxicity (ADMET). Early-stage identification of toxicity risks helps minimize clinical trial failures and ensures patient safety—a critical advantage in regulatory approval processes.
Future Directions of AI in Biopharma

The trajectory of AI-enabled drug discovery continues to evolve. Innovations on the horizon include:
- Generative AI for Molecular Scaffold Design: Leveraging generative algorithms to propose novel chemical scaffolds that optimize pharmacological activity.
- Digital Twins for Drug Development: Creating virtual replicas of biological systems to simulate disease progression and evaluate therapeutic interventions comprehensively.
- Personalized Medicine Integration: Combining genomic data with AI to design individualized therapies tailored to patients’ specific molecular profiles.
Conclusion
As life sciences move boldly into the era of computational biology, AI-driven approaches promise to dismantle longstanding bottlenecks in drug research. By enabling rapid molecule design, iterative testing, and large-scale data analysis, these methodologies are paving the way for precision therapies that enhance patient outcomes. Medvolt’s contributions showcase how interdisciplinary expertise—spanning AI, enzymology, and molecular modeling—can deliver transformative results for biomedical challenges. Whether addressing unmet clinical needs in oncology, rare diseases, or metabolic disorders, AI’s synergy with domain knowledge holds immense promise for the future.
Innovators, investors, and industrial stakeholders alike must seize this moment to harness AI’s potential as we collectively build a more efficient, impactful ecosystem for drug discovery.