Innovations in C–N Bond Formation: Enhancing Amine Synthesis with AI and Green Chemistry
Advances in C–N bond formation are transforming amine synthesis, enabling greener chemistry and AI-driven optimization in drug discovery.
Introduction
The formation of carbon-nitrogen (C–N) bonds is foundational to organic chemistry, forming the backbone of many pharmaceuticals, agrochemicals, and materials. Amine synthesis, in particular, has long been a cornerstone of therapeutic molecule design. Despite decades of advancements, existing challenges in efficiency, specificity, and sustainability still impede the discovery and manufacturing pipelines in drug development.
Recently, a breakthrough in C–N bond formation has emerged, offering new pathways to streamline amine synthesis. This innovation is not merely a chemical advancement—it opens doors for integration with AI-driven platforms to accelerate drug development, optimize reaction conditions, and expand access to novel chemical space.
This article explores the implications of this new method for the biotechnology and pharmaceutical industries, with a focus on its potential synergy with AI-enabled R&D, enzyme engineering, and Medvolt’s capabilities in computational chemistry.
The Challenge: Why C–N Bond Formation Matters
C–N bonds are ubiquitous in almost all drug molecules, ranging from simple small-molecule inhibitors to complex biologics. However, their formation is far from straightforward. Traditional methods for amine synthesis often rely on:
- Harsh reaction conditions, such as high temperatures or toxic reagents.
- Limited substrate scope, leading to inefficiencies in functional group tolerance.
- Low stereoselectivity, which complicates the production of chiral intermediates with high optical purity.
- Sustainability issues, such as waste generation and the environmental impact of catalysts.
Given the highly iterative nature of drug discovery, where thousands of molecular candidates are tested and optimized, inefficiencies in C–N bond formation can magnify bottlenecks. Streamlining this key reaction step could therefore yield profound downstream benefits for lead optimization, scale-up, and ultimately, time-to-clinic.
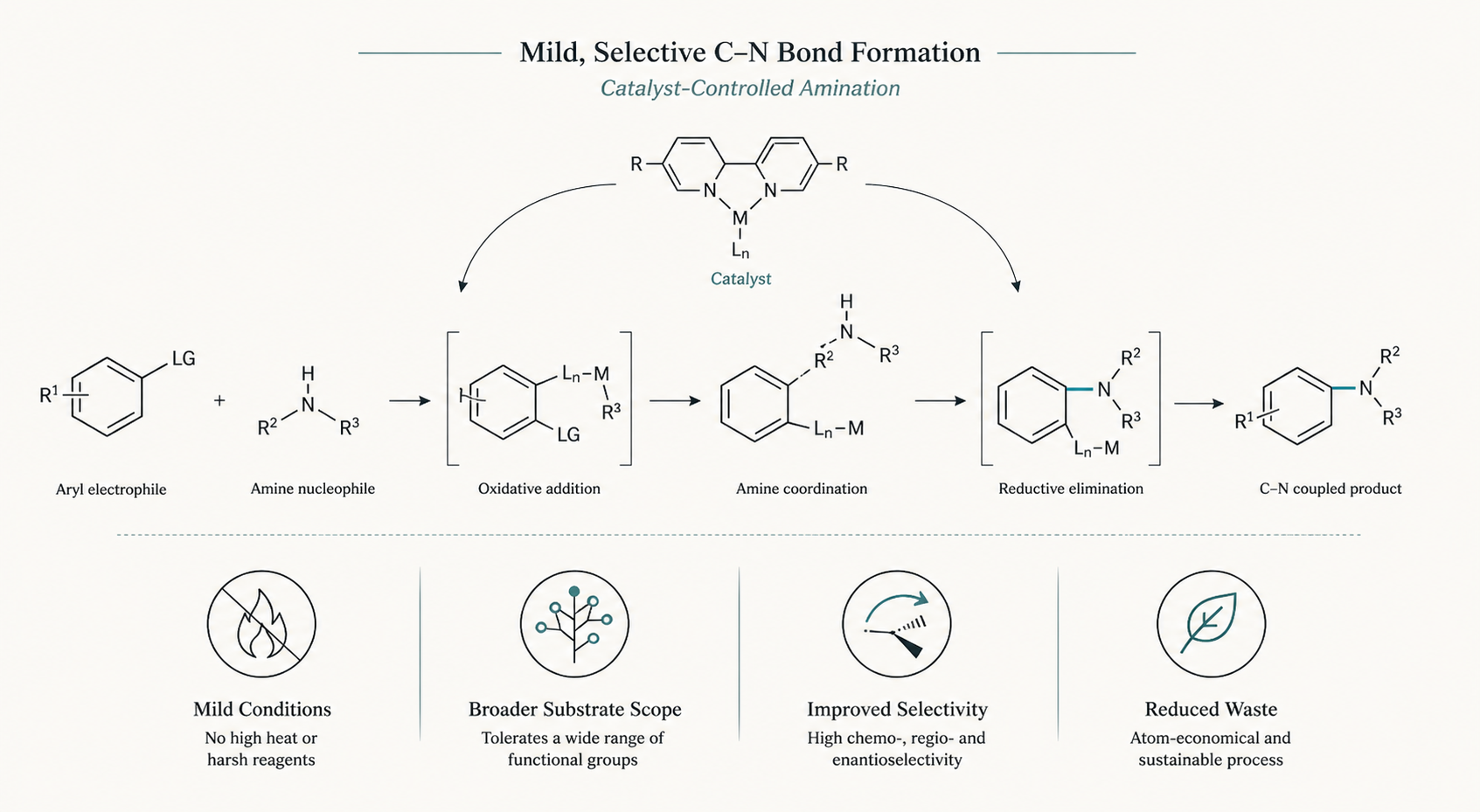
The New Breakthrough in C–N Bond Formation
The latest advancements in C–N bond formation leverage a combination of innovative catalytic strategies and novel reaction pathways. One promising method, for example, involves coupling amines directly with electrophiles under mild, environmentally benign conditions.
Key Advantages:
- Mild Reaction Conditions: The new method minimizes the use of harsh reagents like reducing metals or strong acids. This reduces both costs and environmental impact while improving compatibility with sensitive drug-like molecules.
- Broader Substrate Scope: By employing tailored catalysts, the reaction accommodates a wider variety of functional groups, enabling efficient diversification of molecular scaffolds.
- Improved Selectivity: Enhanced precision in C–N bond formation allows for better control over regioselectivity and chirality—critical factors in pharmaceutical synthesis.
- Reduced Waste: The catalytic process minimizes byproducts and waste streams, aligning with green chemistry principles.
Such advances provide not just incremental improvements but also foster entirely new opportunities for creating amine-based compounds faster and more sustainably.
Implications for Drug Discovery
The ability to form C–N bonds more efficiently has far-reaching implications for the pharmaceutical sector, especially when integrated with recent advances in AI and computational modeling.
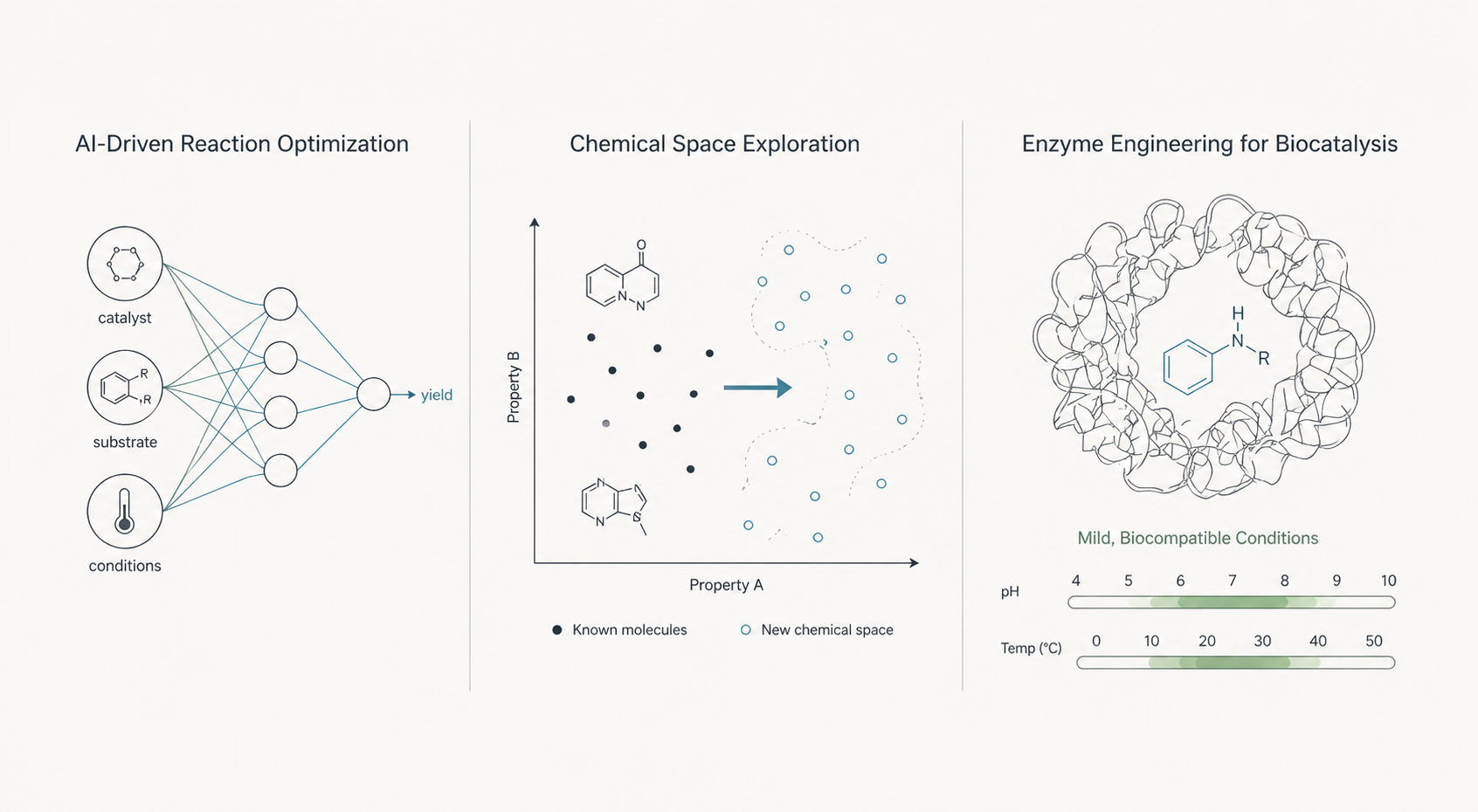
AI-Driven Reaction Optimization: The Role of Machine Learning
Medvolt’s AI-driven platforms can enhance the application of this new C–N bond formation method by rapidly optimizing reaction conditions. Machine learning models, trained on multi-omics datasets and reaction-specific parameters, can predict:
- Optimal catalyst-ligand combinations to improve yield.
- Reaction conditions tailored to novel substrates.
- Environmental variables affecting regioselectivity and chirality.
Through such predictive insights, Medvolt greatly reduces the trial-and-error inherent in traditional medicinal chemistry workflows.
Extending Access to New Chemical Space
Amine synthesis is central to exploring previously inaccessible regions of chemical space. The broader substrate scope of this new method, combined with AI-driven synthesis planning, aids medicinal chemists in:
- Designing analog libraries around existing drug candidates.
- Expanding fragment-based drug designs into lead-like molecules.
- Developing small molecules with specific physicochemical properties for challenging targets (e.g., allosteric modulators).
This ensures that biopharma pipelines remain competitive and innovative at each stage of R&D.
Enzyme Engineering for Biocatalysis
Biocatalysis has increasingly emerged as a green alternative for amine synthesis. By leveraging AI-guided enzyme engineering, Medvolt can identify and design enzymes tailored to specific C–N bond-forming reactions. These engineered enzymes exhibit:
- High activity at mild pH and temperature.
- Substrate promiscuity or selectivity, depending on application needs.
- Scalability for both discovery-phase and commercial production.
Sustainability: Aligning with Green Chemistry
The pharmaceutical industry is under growing pressure to adopt environmentally sustainable practices. Green chemistry principles such as waste minimization, atom economy, and renewable feedstocks are becoming standardized benchmarks along the value chain. This new method for C–N bond formation aligns perfectly with these goals:
- Reduced Reagent Consumption: Less reliance on stoichiometric reagents lowers operational costs and waste.
- Lower Energy Inputs: Mild reaction conditions minimize energy consumption and thermal hazards.
- Scalable Green Catalysts: Broadly applicable catalysts reduce ecological risk.
In combination with AI-designed route prediction software, Medvolt’s platforms enable sustainability metrics to be incorporated directly into process development.
Future Perspectives: Beyond Amine Synthesis
While the current focus centers on amine synthesis, innovations in C–N bond formation have implications across other applications in pharmaceutical and material chemistry. Future potential areas include:
- DNA-Encoded Libraries: Efficient amine coupling reactions can expand the diversity of DNA-tagged small molecules for high-throughput screening.
- Peptide and Protein Modification: Covalent C–N bonds are essential for conjugation chemistries in peptide therapeutics and antibody-drug conjugates (ADCs).
- Biodegradable Polymers: Functional amines are critical in designing biocompatible polymers for drug delivery systems.
Medvolt’s expertise in multi-omics analysis ensures seamless integration of these advancements across diverse domains.
Conclusion
The introduction of a streamlined C–N bond formation protocol marks a significant step forward for the life sciences sector. By addressing long-standing challenges in amine synthesis, this breakthrough enhances the efficiency, sustainability, and versatility of pharmaceutical and chemical R&D pipelines.
With AI-driven tools like those deployed by Medvolt, the adoption of new methodologies can be further accelerated, unleashing rich opportunities for innovation. As the life sciences landscape continues to evolve, such advances reaffirm the critical partnership between computational methods and wet-lab experimentation in driving transformative change.