The €25B vs €219B Problem: Europe's Plan to Revive Biotech with AI and Innovative Policies
Europe’s biotech sector faces a funding and innovation gap, but AI, policy reform, and strategic investment could redefine its global competitiveness.
Introduction
The biotechnology landscape in Europe is at a crossroads. Despite possessing a rich history of scientific innovation, Europe’s life sciences sector lags significantly behind in driving venture capital (VC) investments compared to the United States. Between 2015 and mid-2025, EU biotech startups attracted €25 billion in venture capital funding. In stark contrast, their American counterparts secured €219 billion—a disparity that underscores the challenges Europe faces in translating scientific prowess into commercially viable solutions.

The Root of Europe’s Biotech Problem
While Europe excels in fundamental research, it struggles to convert discoveries into market-ready innovations. Historically, Europe accounted for half of new pharmaceutical treatments worldwide. Today, that share has dwindled to just 20%. The reasons behind this decline are manifold, including:
- Fragmented Regulatory Environment: Operating across 27 countries with distinct legislative frameworks deters startups from scaling efficiently. Regulatory hurdles increase time-to-market and dissuade VC investment.
- Underdeveloped Equity Markets: The limited role of private equity funds in Europe hampers large-scale investments.
- Brain Drain & Market Listings: Sixty-six of the 67 EU biotech companies that went public in the last six years opted for foreign stock exchanges instead of EU markets, signaling a loss of growth potential within the region.
Beyond these systemic challenges, the biotech ecosystem in Europe also trails in adopting frontier technologies, including artificial intelligence (AI). AI’s integration into drug discovery and clinical workflows is seen as a critical enabler for transforming scientific innovation into tangible products. Yet here, too, Europe lags behind.
Europe’s Biotech Ambitions: The European Biotech Act and EU-Inc
Recognizing these gaps, the European Union is enacting a suite of legislative and strategic measures. Leading this charge is the European Biotech Act, a proposal designed to catalyze innovation and investment in Europe’s biotechnology sector. It includes several targeted interventions:
- Simplified Access to Finance: By facilitating better funding channels, particularly for high-risk R&D projects, the Biotech Act addresses early-stage capital gaps.
- Extension of Patent Protections: Eligible biotech medicines developed and manufactured in the EU benefit from extended intellectual property coverage, incentivizing local manufacturing and research activity.
- Streamlined Regulatory Procedures: A directive for the European Medicines Agency (EMA) to redefine clinical trial data requirements for biosimilars could reduce development timelines and costs.
Complementing these measures is a second initiative: EU-Inc, a pan-European corporate structure aimed at reducing administrative complexities. Spearheaded by European Commission President Ursula von der Leyen, EU-Inc promises to allow businesses to register within 48 hours through a fully digital, English-language process. This move directly addresses the compliance burdens faced by startups navigating transnational regulatory systems. By creating a unified corporate framework, policymakers hope to position Europe as a dynamic hub mirroring Silicon Valley’s entrepreneurial ecosystem.
The Role of AI in Europe’s Biotech Revival
One of the most transformative tools in modern biotechnology is artificial intelligence. Yet, Europe’s ability to leverage AI remains constrained by underinvestment and regulatory inertia. In 2024, NVIDIA CEO Jensen Huang and French President Emmanuel Macron drew attention to Europe’s widening gap with the United States and China in AI expenditure and implementation.
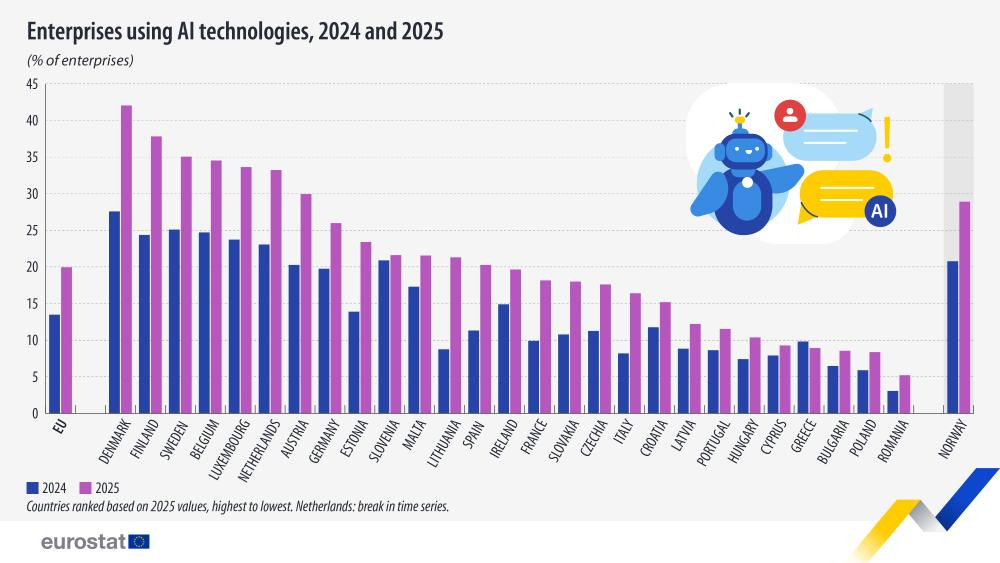
According to the Stanford AI Index Report, the U.S. topped global AI innovation with 40 notable AI models in 2024, compared to 15 in China and just 3 in Europe. Despite these shortcomings, AI adoption in Europe is growing. By 2025, 20% of medium and large enterprises were reportedly using AI—a significant improvement from 7.7% in 2021. The European Commission believes that AI, particularly when applied to drug discovery, diagnostic tools, and predictive modeling, can unlock Europe’s biotech potential.
AI in Drug Discovery and Enzyme Engineering: Reducing the R&D Bottleneck
Biotech innovation is inherently time-intensive, with the average drug development timeline exceeding 10 years and success rates from clinical trials hovering under 15%. AI-driven methodologies, however, are rapidly shifting this paradigm. For instance:
- AI-Assisted Molecular Modeling: Advances in algorithms, including free-energy perturbation (FEP) simulations and fragment-based design, enable researchers to predict molecular interactions with unprecedented speed and accuracy. This can shave years off preclinical development cycles.
- Multi-Omics Integration: AI excels at analyzing vast datasets spanning genomics, proteomics, and metabolomics. These insights are helping researchers identify novel therapeutic targets and biomarkers, especially for complex diseases like cancer and neurodegenerative disorders.
- Precision Design in Enzyme Engineering: AI-powered tools are accelerating protein design to enhance metabolic pathways or produce next-generation biosynthetic enzymes. These innovations stand to revolutionize pharmaceuticals, biomanufacturing, and even renewable energy.
Harnessing AI for Drug Discovery: The Race to Innovate and Govern

Medvolt’s AI-assisted R&D platforms represent a benchmark in enabling precision workflows across these domains. By integrating computation with experimental feedback loops, Medvolt helps overcome key challenges in molecular optimization and biophysical testing, unlocking actionable insights at each stage of development.
Striking the Balance Between Regulation and Innovation
The EU’s dual emphasis on ethical governance—set forth through frameworks like the 2024 AI Act—and innovation often strikes a precarious balance. While well-intentioned, the stringent requirements for safety, traceability, and transparency in AI implementation can inflate costs and impede progress. By comparison, the U.S. operates with looser restrictions, resulting in faster adaptation in industries like biotech and pharmaceutical research.
Europe’s regulatory reform, as envisioned under the Biotech Act, is a strategic attempt to recalibrate this balance. Streamlined guidance for AI applications in biotech—notably in automated drug discovery campaigns—could remove current bottlenecks without compromising ethical safeguards. Lessons from successful AI deployments, such as virtual screening, clinical trial optimization, and robotic labs, can be codified into industry guidelines without stifling creativity.
A Unified Vision for Europe’s Biotech Leadership
The ambition to close the €25B versus €219B gap is not merely about capital flows; it’s about rediscovering Europe’s cutting-edge capacity to develop paradigm-shifting solutions for global health challenges. The proposed Biotech Act, alongside modernized AI frameworks, signals a multi-pronged approach, blending legislative, technical, and financial strategies.
For Europe to establish itself as a leader in AI-powered drug discovery and biotechnology, it must prioritize cross-border collaboration between academia, industry, and government bodies. This includes:
- Investing in AI Infrastructure: Providing computational resources and funding programs for high-throughput, AI-driven experiments.
- Cross-Disciplinary Training for Talent: Equipping scientists with skills in computational chemistry, bioinformatics, and machine learning.
- Creating Public-Private Research Consortia: Driving partnerships to commercialize research outputs more effectively.
By addressing systemic inefficiencies and embracing AI innovation, Europe has a historic opportunity to turn its biotechnology sector into a cornerstone of 21st-century healthcare. Initiatives such as the Biotech Act pave the way, but collaboration, ingenuity, and public trust will be critical components of its long-term success.
Conclusion
Europe’s biotech sector has the intellectual firepower and institutional depth to rival its global peers, yet systemic barriers continue to hold back its potential. Although the €25B to €219B funding disparity mirrors deeper structural challenges, new legislative reforms and AI-driven strategies present an actionable path toward revitalizing the industry.
Companies at the forefront of innovation, like Medvolt, are poised to play an integral role in this transformation by leveraging data and technology to accelerate breakthroughs in drug discovery and biomanufacturing. Europe’s journey to close the gap is not just an economic ambition—it is a chance to redefine the future of healthcare on a global scale.