The Shift from Screening to Designing Molecules: How Drug Discovery Is Moving Beyond Libraries to Intelligent Creation
Drug discovery is moving beyond library screening toward intelligent molecule design powered by generative AI, structural biology, and physics-based validation.
Introduction
For decades, drug discovery has been driven by a simple idea:
Find molecules that work.
The dominant approach was screening, whether through high-throughput experiments or computational filtering of large chemical libraries. The assumption was straightforward:
If you test enough molecules, you will eventually find a hit.
But today, that assumption is being challenged.
The pharmaceutical industry is undergoing a fundamental shift, moving from screening existing compounds to designing new molecules with desired properties from the ground up.
This transition is not just technological.
It is economic, scientific, and strategic.
The Traditional Model: Screening at Scale
Historically, drug discovery relied heavily on high-throughput screening (HTS) and later virtual screening.
These approaches aimed to:
- evaluate millions of compounds
- identify a small subset of hits
- optimize those hits into leads
Virtual screening, in particular, became a computational extension of this paradigm, enabling researchers to evaluate vast libraries of molecules against biological targets.
At its core, screening has always been a numbers game.
The challenge is the size of chemical space.
- Theoretical chemical space exceeds 10⁶⁰ possible molecules
- Even large screening libraries cover only a tiny fraction of this space
This creates a fundamental limitation.
You are not discovering the best molecule.
You are discovering the best molecule within what you already have.
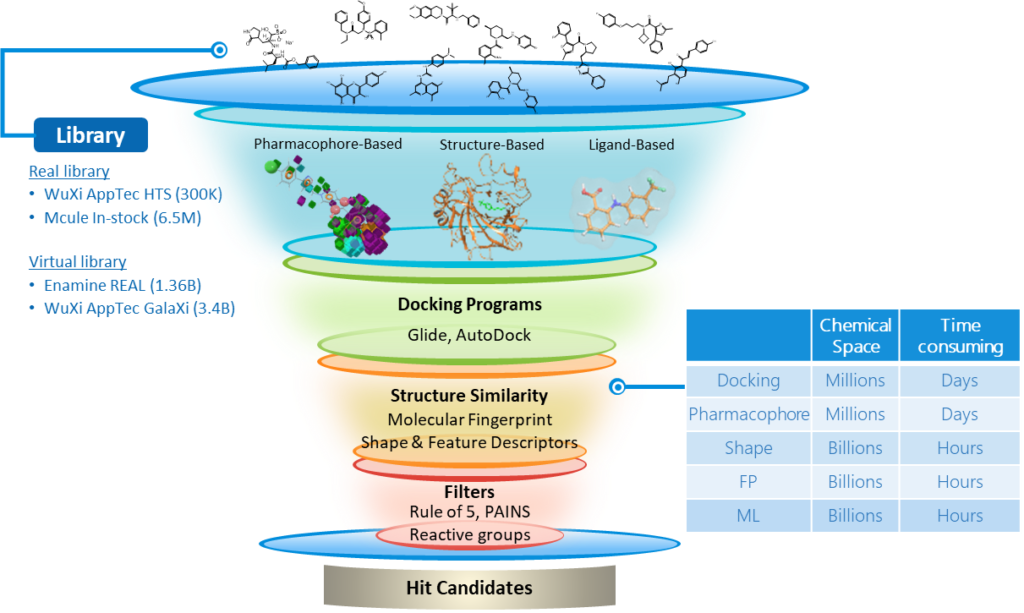
Why Screening Is No Longer Enough
While screening has driven many successful drugs, its limitations are becoming increasingly apparent.
1. Limited Exploration of Chemical Space
Even screening millions of compounds barely scratches the surface of possible drug-like molecules.
This means:
- many potentially optimal molecules are never tested
- innovation is constrained by existing libraries
2. High Cost and Low Efficiency
Screening workflows, especially experimental HTS, are:
- resource-intensive
- time-consuming
- often yield low hit rates
Even virtual screening, while faster, still relies on filtering rather than creating.
3. Poor Early-Stage Decision Quality
Screening identifies hits based on:
- binding affinity
- limited scoring functions
But it often fails to capture:
- ADMET properties
- selectivity
- long-term stability
This leads to high attrition in later stages.
The Emergence of Molecular Design
The new paradigm is fundamentally different.
Instead of asking:
“Which molecules should we test?”
The question becomes:
“What molecule should we design?”
This is the core of de novo drug design, where entirely new molecular structures are created to fit a biological target.
This shift is being enabled by advances in:
- generative AI
- molecular modeling
- structural biology
- computational chemistry
The Role of Generative AI in Molecule Design
Generative AI has introduced a powerful new capability into drug discovery:
The ability to create novel molecules with specific properties.
Modern generative models can:
- learn chemical representations from large datasets
- generate new molecular structures
- optimize multiple properties simultaneously
These models include:
- variational autoencoders (VAEs)
- generative adversarial networks (GANs)
- diffusion models
- transformer-based architectures
This enables a shift from exploration to intent-driven design.
Why Designing Molecules Changes the Economics
The transition from screening to design is not just about better science.
It fundamentally changes the economics of drug discovery.
1. Reduced Search Space
Instead of exploring billions of compounds blindly, researchers can:
- focus on high-probability candidates
- generate molecules tailored to targets
2. Faster Iteration Cycles
Design-driven workflows allow:
- rapid generation of candidates
- immediate computational evaluation
- faster optimization loops
In some cases, AI-driven approaches have been shown to:
- reduce development timelines
- lower costs by up to 50–70% in early stages
3. Improved Success Rates
Early evidence suggests that AI-designed molecules show:
- 80–90% success rates in Phase I trials, significantly higher than historical averages
While still evolving, this indicates that better-designed molecules can translate into better clinical outcomes.
4. Multi-Objective Optimization
Unlike screening, which focuses on a single metric, design allows:
- simultaneous optimization of potency, selectivity, and ADMET
- balancing trade-offs early
This reduces late-stage failures.
The Limitations of Pure Generative Approaches
Despite its promise, generative AI is not a complete solution.
There are key challenges:
1. Dependence on Training Data
Generative models are only as good as the data they learn from.
Poor data leads to:
- unrealistic molecules
- lack of novelty
2. Lack of Physical Validation
AI models can suggest molecules, but they do not inherently understand:
- binding dynamics
- molecular interactions
- thermodynamics
This creates a gap between prediction and reality.
3. Multi-Parameter Complexity
Designing a molecule is not a single-objective problem.
It involves balancing:
- binding affinity
- solubility
- toxicity
- synthesizability
Current models still struggle with fully capturing this complexity.
The Hybrid Model: Design + Physics
The future of drug discovery is not purely generative.
It is integrated.
The most effective workflows combine:
- AI-driven molecule generation
- structure-based modeling
- physics-based simulations
This hybrid approach ensures that:
- generated molecules are realistic
- interactions are physically validated
- predictions are actionable
Generative AI has the potential to significantly accelerate drug discovery, improving efficiency and reducing costs when combined with robust validation frameworks.
Medvolt’s Approach: From Screening to Intelligent Design
At Medvolt, we approach small molecule discovery as a design problem, not a filtering problem.
Our platform, MedGraph, is built around this philosophy.
Generative Design with Constraints
Instead of generating arbitrary molecules, we focus on:
- chemistry-aware generation
- scaffold-driven design
- synthesizability constraints
This ensures that generated compounds are practically viable.
Structure-Aware Optimization
We integrate:
- protein structure modeling
- active-site analysis
- binding pocket characterization
to guide molecule design toward realistic interactions.
AI + Physics Integration
Our workflows combine:
- generative AI for molecule creation
- docking and AI-based rescoring
- molecular dynamics (MD) simulations
- free energy perturbation (FEP)
This ensures that molecules are not just predicted to bind, but are validated for stability and interaction dynamics.
Multi-Parameter Optimization
We evaluate molecules across:
- potency
- selectivity
- ADMET properties
- developability
This reduces downstream risk and improves success probability.
What This Shift Means for the Industry
The transition from screening to designing molecules will reshape the pharmaceutical landscape.
1. From Library Dependency to Creativity
Drug discovery will no longer be limited by:
- available compound libraries
Instead, researchers can explore novel chemical space.
2. From Trial-and-Error to Prediction
Design-driven workflows reduce reliance on:
- blind experimentation
and enable data-driven decision-making.
3. From Incremental to Transformational Innovation
Instead of modifying existing molecules, researchers can:
- create entirely new therapeutic classes
4. Democratization of Drug Discovery
Smaller teams with advanced computational tools can now:
- compete with large pharmaceutical companies
This lowers barriers to innovation.
The Road Ahead
The shift from screening to designing molecules is still ongoing.
Challenges remain in:
- data quality
- model interpretability
- experimental validation
But the direction is clear.
Drug discovery is moving toward a future where:
- molecules are designed with intent
- validated with physics
- optimized across multiple parameters
Conclusion
For decades, drug discovery has been driven by screening.
But screening is inherently limited.
It can only find what already exists.
Design, on the other hand, creates what does not.
This is the fundamental shift shaping the future of pharmaceutical innovation.
At Medvolt, we believe the next generation of drug discovery will not be defined by how many molecules you can test.
It will be defined by how intelligently you can design them.
The question is no longer:
“What can we find?”
It is:
“What should we create?”